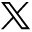Abstract
BACKGROUND AND OBJECTIVES: The pathophysiology of saccular aneurysms is complex and multifactorial. The aim of the present study was to understand the mechanism of apoptosis in an elastase-induced aneurysm model in rabbits.
MATERIALS AND METHODS: Elastase-induced saccular aneurysms were created at the origin of the right common carotid artery in 20 rabbits. Aneurysm samples were harvested at 2 and 12 weeks after creation. Expression of apoptosis-associated proteins, including caspases and bcl-2 proteins, were assessed by Western blot analysis (n = 5 at both time points). Terminal deoxynucleotidyltransferase–mediated dUTP nick end-labeling (TUNEL) staining, which indicates the presence of apoptosis, was performed in tissue sections (n = 5 at both time points). The unoperated contralateral common carotid artery was used as a control.
RESULTS: Expression of active caspase-3, the final executioner of apoptosis, and caspase-9, the mediator of the intrinsic mitochondrial pathway, was observed in aneurysms at 2 weeks, whereas the expression of activated caspase-8, the mediator of the extrinsic death receptor pathway, was absent at both time points. Expression of antiapoptotic proteins, Bcl-2 and phospho-Bad, was down-regulated in aneurysms compared with controls at 2 weeks. None of these proteins were differentially expressed at 12 weeks. These results were confirmed by the presence of TUNEL-positive cells in some aneurysms at the early time point.
CONCLUSIONS: In this study of elastase-induced aneurysms in a rabbit model, activation of apoptosis is mediated predominantly by the Bcl-2-mediated intrinsic pathway through the activation of caspase-9.
Cerebral saccular aneurysms are characterized by profound structural and functional remodeling within the vessel wall, in some cases, associated with progressive arterial dilation and eventual rupture. Most vascular remodeling procedures, however, are adequate and prevent dilation and eventual rupture.1,2 Although the exact pathogenesis remains unknown, several environmental and genetic factors that may lead to continued irreversible destruction of the vessel wall have been recognized.3,4
Apoptosis is an energy-dependent form of cell death that occurs in a wide range of physiologic conditions, such as tissue remodeling, cell proliferation, and tumor regression.5–7 It is characterized by cytoplasmic and nuclear condensation, DNA fragmentation, membrane blebbing, and engulfment and lysosomal degradation of apoptotic bodies by phagocytosis. Apoptosis can be initiated by either the extrinsic pathway, intrinsic pathway, or both, leading to the activation of the final executioner, caspase-3, which results in fragmentation of the nucleus.8,9
We have developed an experimental elastase-induced aneurysm model in the rabbit carotid artery, which structurally mimics human intracranial saccular aneurysms.10–12 We previously reported the occurrence of apoptosis in the aneurysm sac after embolization with platinum coils.13,14 In the present study, we aimed to evaluate the presence of apoptosis at different stages of aneurysm growth of the elastase-induced experimental aneurysm.
Materials and Methods
Aneurysm Creation
The Institutional Animal Care and Use Committee approved all procedures before initiation of the study. Elastase-induced saccular aneurysms were created in 20 New Zealand white rabbits (body weight, 3–4 kg) by using the rabbit-elastase model. Detailed procedures for aneurysm creation have been described in depth elsewhere.15 Briefly, anesthesia was induced with an intramuscular injection of ketamine, xylazine, and acepromazine (75, 5, and 1 mg/kg, respectively). Using a sterile technique, we exposed the right common carotid artery (RCCA) and ligated it distally. A 1- to 2-mm beveled arteriotomy was made, and a 5F vascular sheath was advanced retrograde in the RCCA to a point approximately 3 cm cephalad to the origin of RCCA. A 3F Fogarty balloon was advanced through the sheath to the level of the origin of the RCCA with fluoroscopic guidance and was inflated with iodinated contrast material. Porcine elastase (Worthington Biochemical, Lakewood, New Jersey) was incubated within the lumen of the common carotid artery above the inflated balloon for 20 minutes, after which the catheter, balloon, and sheath were removed. The RCCA was ligated below the sheath entry site, and the incision was closed. Aneurysms were allowed to mature either for 2 weeks (n = 5 each for molecular biology and terminal deoxynucleotidyltransferase [TdT]−mediated dUTP nick end-labeling [TUNEL] assay) or for 12 weeks (n = 5 each for molecular biology and TUNEL assay).
Tissue Harvest
Aneurysm samples were harvested at either 2 weeks (n = 10) or 12 weeks (n = 10) following aneurysm creation. Under general anesthesia, animals were euthanized by a lethal injection of sodium pentobarbital. The aneurysm and unoperated left common carotid artery (LCCA) were then harvested. The samples were placed in buffered formalin for TUNEL staining. If the aneurysms were relegated to molecular biology experiments, the aneurysm samples were dissected into 2 samples, including the neck and dome. These samples were kept frozen at −70°C.
TUNEL Staining
TUNEL staining was performed for specific labeling of nuclear DNA fragmentation, an important biochemical hallmark of apoptosis, with the DeadEnd Fluorometric TUNEL system (Promega Corp, Madison, Wisconsin). With this method, apoptosis can be quantified at the single-cell level; however, it does not detect the apoptotic cells at their early stages. The fixed vessels were dehydrated in an ascending series of ethanol, cleared in xylene, and embedded in paraffin. The paraffin-embedded tissues were then sectioned at a 5-μm thickness. The tissue sections were deparaffinized and rehydrated, followed by fixing them in 4% paraformaldehyde for 15 minutes. They were incubated with 20 μg/mL of proteinase K for 20 minutes at room temperature, and then sections were rinsed with phosphate buffered solution (PBS) and kept in the equilibration buffer for 10 minutes at room temperature. They were incubated with 100 μL of TdT reaction (rTdT) mix containing the rTdT enzyme and fluorescent-labeled nucleotides in a humid atmosphere for 1 hour at 37°C. The reaction was terminated by immersing the slides in water for 15 minutes. The sections were then immersed in 2× SSC buffer for 15 minutes at room temperature to terminate the reaction and then were rinsed with PBS. The slides were analyzed by fluorescence confocal microscopy. Positive controls consisted of incubating sections with deoxyribonuclease I. A negative control was performed by incubating the section with rTdT reaction mix without the rTdT enzyme.
Immunoblotting
Frozen samples were pulverized under liquid nitrogen and extracted in an ice-cold lysis buffer (10-mmol/L sodium phosphate, pH 7.2, 150-mmol/L sodium chloride, 1% Triton X-100, 0.1% sodium dodecyl sulfate [SDS], 0.5% sodium deoxycholate, and 0.2% sodium azide with a protease inhibitor cocktail). After centrifugation at 10,000 g for 20 minutes at 4°C, the protein concentration of the supernatant was determined (Pierce Biotechnology, Rockford, Illinois).
Total protein was fractionated on SDS polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane (Bio-Rad, Hercules, California). Membranes were incubated with monoclonal anti-bcl-2, phospho-Bad, caspase-8 (Cell Signaling Technology, Danvers, Massachusetts), rabbit polyclonal anti-caspase-3, and caspase-9 (Cell Signaling Technology) overnight at 4°C. The membranes were incubated with the appropriate secondary antibody; either a sheep anti-rabbit or goat anti-mouse immunoglobulin G (Bio-Rad) dilution conjugated to horseradish peroxidase. Immune complexes were visualized by using the enhanced chemiluminescence detection system (ECL Western Blotting Detection Reagents; GE Healthcare, Piscataway, New Jersey), and bands were measured by densitometric analysis of autoradiograph films (UN-SCAN-IT software, Silk Scientific, Orem, Utah).
Statistical Analysis
Results are expressed as mean ± SD. The unoperated LCCAs were used as controls. Differences between groups were assessed by the Student t test. Statistical significance was taken as a probability value of <.05. Each value was compared with the LCCA of the same time period, as a control. The values from the 2-week group and 12-week group were also compared.
Results
TUNEL Staining
The TUNEL assay was applied on tissue sections to detect apoptotic cells. At 2 weeks after aneurysm creation, 2 of 5 aneurysms (40%) were positive for TUNEL staining (Fig 1). No TUNEL-positive cells were observed in any of the 12-week aneurysms.
Terminal deoxynucleotidyltransferase−mediated dUTP nick end-labeling (TUNEL) staining. Immunofluorescence image of TUNEL staining at the distal neck of a 2-week aneurysm (original magnification ×150).
Expression of Caspases
The expression of caspases was analyzed in control and aneurysm samples by using Western blotting. There was no difference in expression between aneurysms and controls for pro-caspase-3 (35 kDa), pro-caspase-8 (57 kDa and pro-caspase-9 (35 kDa) at either time point. Expression of cleaved activated-caspase-3 (17 kDa) and caspase-9 (17 kDa) was detected in the neck and dome of aneurysms compared with controls at 2 weeks but not at 12 weeks (P < .001) (Fig 2).
Expression of proapoptotic proteins. A, Representative Western blot images of the expression of caspase family proteins from rabbits at 2 weeks and 12 weeks post-aneurysm creation. B, Densitometric analysis of the expression of procaspase-3 between control and experimental animals. C, Densitometric analysis of the expression of cleaved-caspase-3 between control and experimental animals. D, Densitometric analysis of the expression of procaspase-8 between control and experimental animals. E, Densitometric analysis of the expression of procaspase-9 between control and experimental animals. F, Densitometric analysis of the expression of cleaved-caspase-9 between control and experimental animals. LCCA indicates left common carotid artery; PA, parent artery; AN, aneurysm neck; AD, aneurysm dome; a, P < .05 versus control; b, P < .05 versus the corresponding 2-week group.
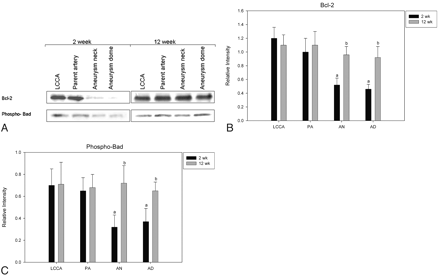
Expression of bcl-2 and Phospho-Bad
Because the cleavage of pro-caspase-9 to active cleaved caspase is associated with the expression of antiapoptotic proteins bcl-2 and phospho-Bad, we analyzed the expression of those proteins in the aneurysm samples. The levels of both bcl-2 and phospho-Bad were significantly lower in aneurysms at 2 weeks (P < .001) compared with controls (Fig 3). At 12 weeks, the expressions of bcl-2 and phospho-Bad were not significantly different between aneurysms and controls.
Expression of antiapoptotic proteins. A, Representative Western blot images of the expression of bcl-3 and phospho-Bad from rabbits at 2 weeks and 12 weeks post-aneurysm creation. B, Densitometric analysis of the expression of bcl-2 between control and experimental animals. C, Densitometric analysis of the expression of phospho-Bad between control and experimental animals.
Discussion
Development and growth of cerebral saccular aneurysms involves a complex remodeling of cells in the arterial wall. Apoptosis of vascular smooth muscle cells has been widely reported in the pathogenesis of atherosclerosis and abdominal aneurysms.16,17 Kondo et al,18 in a rat model of early cerebral aneurysm development, identified apoptotic medial smooth muscle cells, prompting those authors to posit an association between apoptosis and aneurysm formation.19 The current study used a different animal model than the one in Kondo et al, in which saccular dilation of the proximal RCCA was achieved by distal ligation and proximal elastase injury. Thus, we studied the apoptotic activity of “formed” aneurysms to determine whether programmed cell death occurred after aneurysm formation.
Our results indicated an increase in apoptotic activity and a decrease in antiapoptotic activity early after aneurysm creation. Although not specifically studied in the current work, we have previously shown that our experimental aneurysms grow for approximately 3 weeks following creation. It is possible that ongoing aneurysm growth is promoted by apoptotic activity within the vessel wall, activity that diminishes with time and explains late stability of aneurysm size.
The mechanism that simulated apoptosis in our model remains unknown. Altered shear stress, which is markedly diminished in the aneurysm cavity,20 may lead to apoptosis of the aneurysm wall.21 Direct injury by the elastase may be responsible for the apoptosis. One might study this by adding a control group without elastase, but the vessels in that type of model simply involute.
Activation of caspases (cysteine aspartate-specific proteases), a family of cysteine proteinases, plays a central role in the process of apoptosis. The apoptotic caspases are classified as initiators or executioners, depending on their point of entry into the apoptotic cascade. The initiator caspases are the first to be activated in a particular death pathway, and they constitute the first step in a minimal 2-step cascade by activating the executioner caspases. Procaspases are synthesized as inactive zymogens, which are activated by cleavage.
Apoptosis can be initiated by either an extrinsic and/or intrinsic pathway, both leading to a final common pathway in which activated downstream caspases are involved in DNA fragmentation. The extrinsic pathway, known as the death-receptor pathway, is mediated by the binding of ligands to receptors in the cell surface, which leads to the activation of caspase-8. In the mitochondrial intrinsic pathway, release of cytochrome c from the mitochondria to the cytosol forms an apoptotic complex, which in turn activates the apical caspase of the intrinsic pathway, caspase-9. Activation of caspase-8 and/or caspase-9 results in subsequent activation of the executioner caspase, caspase-3. This then leads to DNA fragmentation and death of the cell.
In the present study, cleaved-caspase-3 and cleaved-caspase-9 were detected at the early time point, while cleaved-caspase-8 was absent; this difference suggests that the apoptotic mechanism is mainly initiated by the intrinsic pathway. Both receptor- and mitochondrial-mediated apoptosis have been previously reported in intracranial aneurysms in humans.22,23 Up-regulation of the caspase-3 gene is closely implicated in the pathologic process of the ruptured intracranial aneurysm.23 Apoptosis in intracranial aneurysms is correlated with increased expression of phosphorylated c-Jun amino-terminal kinase and phosphorylated c-Jun.24
To further confirm the involvement of the mitochondrial pathway in the apoptosis of the elastase-induced aneurysm model, we measured the levels of antiapoptotic proteins, bcl-2 and phospho-Bad, which are associated with the intrinsic pathway. The levels of bcl-2 and phospho-Bad were found to be decreased in 2-week aneurysm samples only. A decreased level of bcl-2 protein lowers the mitochondrial membrane permeability, which leads to the release of cytochrome c to cytosol, the recruitment and activation of caspase-9.
The increased extent of the apoptotic mechanism soon after aneurysm creation indicates that apoptosis may play an important role in the progression of an aneurysm. Previous studies have shown that the endothelial cells along the wall of elastase-induced aneurysms were present at 2 weeks after creation and that the cells were dispersed at 12 weeks.20
Limitations of the Study
TUNEL-positive cells were identified in only 2 of 5 aneurysms after 2 weeks. TUNEL staining detects nuclear DNA strand breaks that happen at the final stages of apoptosis. TUNEL staining does not detect the initial stages of apoptosis. Another limitation is the plane of the histologic section, which does not contain all cells undergoing apoptosis. We analyzed the apoptosis at only 2 time points after creation. In the present study, the LCCA was used as the control, but we acknowledge that a sham-operated control might offer additional valuable data.
Footnotes
-
This study was supported in part by the funds from National Institutes of Health grant (2R01NS042646-04) and the American Heart Association postdoctoral fellowship (AHA0620016Z).
Indicates open access to non-subscribers at www.ajnr.org
References
- Received February 4, 2009.
- Accepted after revision June 17, 2009.
- Copyright © American Society of Neuroradiology