Abstract
BACKGROUND AND PURPOSE: Previous neuropathologic studies in chronic hydrocephalus have suggested the presence of white matter damage, presumably from mechanical pressure due to ventricular enlargement and metabolic derangement. This study aimed to investigate the diffusional properties of the CST in patients with iNPH by using DTI and to determine whether this method could be used as a new diagnostic tool to differentiate patients with iNPH from those with AD and PDD and control subjects.
MATERIALS AND METHODS: We enrolled 18 patients with iNPH, 11 patients with AD, 11 patients with PDD, and 19 healthy control subjects. Diffusion tensor metrics of the segmented CST, including FA values, axial eigenvalues, and radial eigenvalues, were evaluated by using tract-specific analysis. The anisotropy color-coding tractography of the CST was visually evaluated. The DTI findings were compared among groups.
RESULTS: Tract-specific analysis of the CST showed that FA values and axial eigenvalues were significantly increased (P < .001), whereas radial eigenvalues were not significantly altered, in patients with iNPH compared with other subjects. The CST tractographic images in patients with iNPH was visually different from those in other subjects (P < .001). In discriminating patients with iNPH from other subjects, the CST FA values had a sensitivity of 94% and specificity of 80% at a cutoff value of 0.59.
CONCLUSIONS: Our results suggest that patients with iNPH have altered microstructures in the CST. Quantitative and visual CST evaluation by using DTI may be useful for differentiating patients with iNPH from patients with AD or PDD or healthy subjects.
Abbreviations
- AD
- Alzheimer disease
- CST
- corticospinal tract
- DTI
- diffusion tensor imaging
- FA
- fractional anisotropy
- iNPH
- idiopathic normal pressure hydrocephalus
- MMSE
- Mini-Mental State Examination
- PD
- Parkinson disease
- PDD
- Parkinson disease with dementia
- ROI
- region of interest
- SLF
- superior longitudinal fasciculus
iNPH is a syndrome characterized by the triad of gait disturbance, urinary incontinence, and cognitive decline in the absence of elevated CSF pressure.1 Typical features of iNPH on MR imaging studies include ventricular enlargement, diminution of high-convexity sulci, and periventricular hyperintensity.1,2 Previous neuropathologic studies of chronic hydrocephalus have suggested the presence of white matter damage,3–5 presumably resulting from mechanical pressure due to ventricular enlargement6 and metabolic derangement.7
Gait disturbance is the most frequent and reversible symptom in iNPH patients.8 Although the origin of gait disturbance is not entirely understood, various hypotheses, including deformation of the CST, have been proposed.1 The CST connects the motor cortex and spinal cord via the cerebral peduncle. It runs along the side of the lateral ventricle and is presumably deformed by ventricular enlargement. Therefore, we hypothesized that the microstructure of the CST in iNPH patients is altered by mechanical pressure resulting from ventricular enlargement.
Recently, DTI has been used frequently to evaluate white matter alteration in various neurodegenerative diseases.9,10 Diffusion anisotropy is most commonly quantified by the FA value.11 FA values are calculated from the axial and radial diffusivity of water molecules.12 Axial diffusivity is defined by the axial eigenvalue (λ1 value), ie, the mean magnitude of water diffusion parallel to the axon. Radial diffusivity is defined by the radial eigenvalue (average value of λ2 and λ3), ie, the mean magnitude of water diffusion perpendicular to the axon. Tract-specific analysis is 1 method of DTI data analysis. It uses diffusion tensor tractography as an anatomic landmark, and diffusion tensor metrics are calculated along the drawn tractographic image. The CST is 1 of the targets most frequently evaluated by using tract-specific analysis in various diseases.13,14
From a clinical viewpoint, iNPH is considered a reversible cause of dementia and gait disturbance in older adults because it can be treated by shunt surgery. However, it is often difficult to make a diagnosis of iNPH. Although it is a rare disease, iNPH needs to be differentiated from other common diseases causing dementia, gait disturbance, or both. In terms of dementia, iNPH should be differentiated from AD, the most common cause of dementia. Patients with late-phase AD have diffuse cerebral atrophy, leading to secondary ventricular enlargement.15 In terms of gait disturbance, iNPH should be differentiated from PD. Whereas the gait of patients with iNPH is typically described as shuffling, “magnetic,” and wide-based,8 it sometimes resembles that of patients with PD, who have a stooped posture and take small shuffling steps with a freezing gait.16 In addition, patients with PD have a high risk of developing dementia,17 and patients with PDD also have diffuse cerebral atrophy, leading to secondary ventricular enlargement.18 Therefore, biologic markers that can differentiate iNPH from AD or PDD are needed.
Here, we aimed to elucidate the diffusional properties of the CST in patients with iNPH by using tract-specific analysis and to determine whether this method could be used as a new diagnostic tool to differentiate patients with iNPH from those with AD and PDD and healthy control subjects.
Materials and Methods
Subject Selection
Subjects were prospectively recruited from patients treated at Kamagaya General Hospital in Chiba, Japan. All subjects were medically interviewed and were given general and neurologic examinations, including the MMSE, by neurologists (T.H. and T.Y.). Information on complaints of memory loss or other subjective cognitive deficits was gathered by means of a caregiver-based interview for all patients.
Inclusion criteria were defined as follows: iNPH was diagnosed according to the clinical diagnostic criteria of probable iNPH.1 AD was diagnosed according to the National Institute of Neurologic and Communicative Disorders and Stroke and the AD and Related Diseases Association criteria for a diagnosis of probable AD.19 PD was diagnosed according to the clinical diagnostic criteria of the Parkinson's Disease United Kingdom Brain Bank.20 PDD was diagnosed according to the clinical diagnosis criteria for probable PDD.17 Normal control subjects were selected on the basis of a normal result in the MMSE and a normal neurologic examination.
Exclusion criteria were defined as follows: Subjects with a history of neurologic diseases such as stroke and epilepsy or with underlying diseases that potentially affected the brain (eg, uncontrolled hypertension or chronic kidney disease) were excluded.
In addition, subjects who had an Evans index between 0.3 (according to the diagnostic criteria for iNPH1) and the maximum Evans index score in non-iNPH subjects (ie, patients with AD or PDD and control subjects) were selected and classified into subgroups. We thus compared the non-iNPH subgroups with the iNPH subgroup under conditions in which all subjects had similar extents of ventricular enlargement.
This study was approved by the institutional review board of Kamagaya General Hospital, and written informed consent was obtained from all subjects.
Staging of Leukoaraiosis
The extent of periventricular white matter T2 hyperintensities (ie, leukoaraiosis) was rated for all subjects according to the Fazekas classification.21
DTI Data Acquisition
DTI data were obtained with a 1.5T clinical scanner (Signa Excite; GE Healthcare, Milwaukee, Wisconsin) with a single-shot echo-planar sequence (TR = 14,000 ms, TE = 103.5 ms, b = 1000 s/mm2, 13 different axes of motion-probing gradients, FOV = 280 mm, matrix = 128 × 128 [interpolated image matrix size, 256 × 256], section spacing = 3 mm, averaging = 2, number of sections = 50, and total acquisition time = 7 minutes).
Tractography and Tract-Specific Analysis of CST
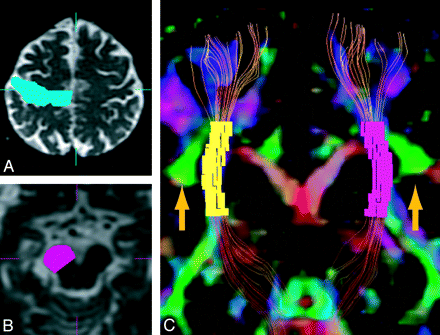
DTI data were analyzed by using dTV II and VOLUME-ONE 1.72, developed by Masutani et al11 (http://www.ut-radiology.umin.jp/people/masutani/dTV.htm) and running on Windows XP Professional (Microsoft, Redmond, Washington). The threshold of line-tracking was set at FA > 0.18. Diffusion tensor tractography of the CST was generated by using a 2-ROI method, with the seed ROI on the motor cortex and the target ROI on the cerebral peduncle (Fig 1A, -B). The CST crosses the SLF at the level of the corona radiata.22 The SLF is identified as the green areas next to the CST on a color-coded FA map of the coronal section in which the CST crosses the SLF (Fig 1C). As an anatomic landmark for the CST, we used the uppermost part of the SLF instead of the lateral ventricle used in a previous study,13 on the basis of the assumption that the areas where the CST and the SLF cross are not affected by the presence or absence of ventricular enlargement (Fig 1). Voxelization along the CST was performed from the uppermost part of the SLF to the posterior limb of the internal capsule by using dTV (Fig 1). The level of the posterior limb of the internal capsule also was identified at one-third of the height from the cerebral peduncle to the uppermost part of the SLF. We evaluated this area in the CST because this area of tractography of the CST was stably drawn. The mean values of the diffusion tensor metrics (ie, FA values, axial eigenvalues, and radial eigenvalues) were measured in the voxelized area of tractography of the CST by using tract-specific analysis, and were averaged between the right and left hemispheres.
Voxelization of part of the CST by tract-specific analysis. Tractographic images of the CST were generated by using the 2-ROI method of seed ROI in the motor cortex (A) and target ROI in the cerebral peduncle (B). C, Tractography of the CST is shown with a color-coded FA map (coronal section). Voxelization along the CST was performed between the uppermost part of the SLF (green area, arrow) and the posterior limb of the internal capsule by using dTV software.
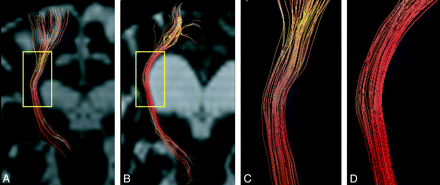
The tractographic colors were visualized by diffusion anisotropy coded from yellow (low anisotropy) to orange (high anisotropy).11 We visually evaluated the same segment of the CST in a tract-specific analysis (Fig 2 A, -B). To avoid any bias resulting from knowledge of ventricular enlargement, the lower and upper parts of the tractography and images of the brain and CSF were not used for visual evaluation. The visual tractographic features of the CST were classified as “type A” (low anisotropy and rough) or “type B” (high anisotropy and straight; Fig 2C, -D) by a neurologist (T.H.) and a neuroradiologist (S.A.) blinded to the clinical information.
Visual evaluation of anisotropy color-coding tractography of the CST. Anisotropy color-coding tractography of the CST in a control subject (A) and a patient with iNPH (B) is shown, with b = 0 images. The areas defined by the yellow squares in A and B were used for visual evaluation in C and D. C, Type A (low anisotropy and rough). D, Type B (high anisotropy and straight).
When creating the tractographic image, the operator (R.S.) was blinded to the clinical information on each subject to avoid bias.
Statistical Analysis
Statistical analysis among groups was performed by using analysis of variance with the post hoc Tukey honestly significant difference test for continuous variables, the Kruskal-Wallis test with post hoc Mann-Whitney U tests for noncontinuous variables, and the χ2 test for categorical data. The tractographic features (the proportion of type B) were compared among groups by using the χ2 test with a post hoc Pearson χ2 test. The criterion of statistical significance was P < .05.
Receiver operating characteristic analysis was used to calculate the optimal cutoffs of the FA value and axial eigenvalue in the CST for patients with iNPH to differentiate their tracts from those of patients with AD or PDD or of control subjects. Analyses were performed with Scientific Package for Social Sciences, Version 11 (SPSS, Chicago, Illinois). The results are expressed as means ± SD.
Results
Demographic and Clinical Features
On the basis of the above-mentioned inclusion and exclusion criteria, we enrolled 18 patients with iNPH, 11 patients with AD, and 11 patients with PDD, as well as 19 age- and sex-matched control subjects (Table 1). All patients with iNPH had cerebral ventricular enlargement (Evans index >0.3) and narrowed CSF spaces at high convexity on MR imaging. The demographic and clinical features of patients with iNPH, AD, or PDD and of control subjects are listed in Table 1. The Evans index was significantly higher in patients with iNPH than in other subjects (P < .001). All patient groups had significantly lower MMSE scores than control subjects (P < .001). There were no significant differences in age, sex, or vascular risk factors (eg, hypertension, diabetes mellitus, and dyslipidemia) among the groups.
Demographic and clinical data of patients with iNPH, AD, or PDD and control subjects
Extent of Leukoaraiosis
The Fazekas classifications of leukoaraiosis for all groups are listed in Table 2. The extent of leukoaraiosis was significantly greater in patients with iNPH than in patients with PDD (P = .035) or control subjects (P = .01). The extent of leukoaraiosis was not significantly different between patients with iNPH and patients with AD (P = .238).
Tract-specific analysis and visual evaluation of tractography of the CST
Diffusion Tensor Metrics in CST of Patients with iNPH, AD, or PDD and in Control Subjects
The diffusion tensor metrics in the CST, as measured by using tract-specific analysis, are listed in Table 2. FA values (P < .001) and axial eigenvalues (P < .001) were significantly increased in patients with iNPH, whereas radial eigenvalues were not significantly altered, compared with those in other subjects.
Table 3 summarizes the diagnostic discrimination of iNPH subjects from the other groups by using FA values and axial eigenvalues of the CST.
Diagnostic discrimination of iNPH patient group from other subject groups
Visual Evaluation of Tractographic Images of CST
Visual evaluation of the tractographic images of the CST showed a greater proportion of type B in patients with iNPH (94.4%) than in patients with AD (27.2%) and PDD (9.1%), or in control subjects (5.3%; Table 2). The proportion of type B was significantly higher in patients with iNPH than in patients with AD or PDD or in control subjects (P < .001). There was sufficient agreement between the assessments made of the tractographic pattern by 2 raters (agreement rate, 52 [88.1%] of 59 subjects) and between 2 assessments made by 1 rater with a 3-month interval between ratings (agreement rate, 54 [91.5%] of 59 subjects).
Comparison of Non-iNPH and iNPH Subjects with Similar Extents of Ventricular Enlargement
The maximum Evans index of our non-iNPH subjects was 0.35. Therefore, subjects who had Evans indexes within the range of 0.3 to 0.35 (ie, 7 patients with iNPH, 4 patients with AD, 6 patients with PDD, and 6 control subjects) were enrolled in this part of the study. There were no significant differences in age, sex, vascular risk factors, Evans index (iNPH, 0.32 ± 0.02; AD, 0.33 ± 0.02; PDD, 0.32 ± 0.02; control, 0.32 ± 0.02; P = .263), or radial eigenvalues (iNPH, 0.54 ± 0.05; AD, 0.55 ± 0.15; PDD, 0.52 ± 0.02; control, 0.55 ± 0.04; P = .888) among all subgroups. FA values were significantly higher in patients with iNPH (0.62 ± 0.02) than in patients with AD (0.56 ± 0.02; P = .009) or PDD (0.58 ± 0.02; P = .038), or in control subjects (0.55 ± 0.04; P = .001). Axial eigenvalues were also significantly higher in patients with iNPH (1.66 ± 0.09) than in patients with AD (1.51 ± 0.05; P = .025) or PDD (1.50 ± 0.09; P = .006), or in control subjects (1.50 ± 0.04; P = .005).
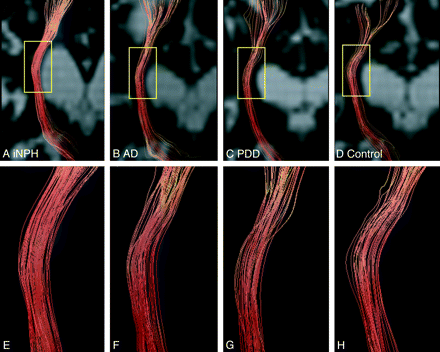
Examples of tractographic images of the CST are shown in Fig 3. Visual evaluation of the tractographic images of the CST showed a greater proportion of type B in patients with iNPH (85.7%), AD (25.0%), and PDD (16.7%) than in control subjects (16.7%). The proportion of type B was significantly higher in patients with iNPH than in patients with AD (P = .044) or PDD (P = .013) or in control subjects (P = .013).
Examples of anisotropy color-coding tractography of the CST. Anisotropy color-coding tractography of the CST in patients with iNPH (Evans index: 0.31; A), AD (0.33; B), PDD (0.33; C), and a control subject (0.31; D) are shown, with b = 0 images. The areas defined by the yellow squares in A, B, C, and D are shown in E, F, G, and H, respectively.
Discussion
Recently, DTI has been applied frequently to the examination of various neurologic diseases.9,10 It is a sensitive tool for detecting subtle microstructural alterations in the cerebral white matter.23 Although FA values decrease in most disease and aging processes, this study showed increased FA values in the CST of patients with iNPH. Increased FA values in the CST also were reported in a previous DTI study of iNPH that used ROI and voxel-based analysis.24 However, this is the first study that quantified the diffusion tensor metrics, including FA values and axial and radial eigenvalues, of the CST by using tract-specific analysis and visually evaluated CST tractography in patients with iNPH; our study then compared the findings with those in patients with AD or PDD and in control subjects.
In our study, patients with iNPH had significantly more extensive leukoaraiosis than patients with PDD or control subjects. In general, the leukoaraiosis indicates structural white matter damage such as myelin loss and ischemia,25 which usually decreases FA values. However, in patients with iNPH, the leukoaraiosis was distributed predominantly around the anterior horn or posterior horn of the lateral ventricle, and it was very limited in extent in the area around the segmented CST in the tract-specific analysis. Thus, we presume that there are little effects of leukoaraiosis for the segmented CST areas.
Here, tract-specific analysis showed that the FA values and axial eigenvalues in the CST were significantly increased, whereas radial eigenvalues were not significantly altered, in patients with iNPH compared with control subjects. In general, FA values are positively correlated with axial eigenvalues and negatively correlated with radial eigenvalues.12 Therefore, the increased FA values in part of the CST in patients with iNPH are due mainly to the increase in axial eigenvalues. As an explanation for these results, we speculate as follows: the brain parenchyma of patients with iNPH is stretched and compressed by mechanical pressure from ventricular enlargement, resulting in stretching of the neural fibers that microscopically show reduced winding and a tendency to align in the direction of stretch. In this altered microstructure, water diffusivity parallel to the axon is enhanced, leading to an increase in axial eigenvalues.
In addition, there might be another explanation for increased FA values. The CST crosses the SLF as well as interhemispheric fibers at the level of the corona radiata.22 In general, FA values decrease at points where fibers cross,11 and they increase when the crossing fibers are lost and only the fibers of interest are left. Interhemispheric fibers pass through the corpus callosum. Mechanical pressure due to ventricular enlargement stretches and compresses the corpus callosum in iNPH,26,27 and this may result in axonal loss and Wallerian degeneration of the interhemispheric fibers. In fact, one study has shown decreased FA values in the corpus callosum in patients with iNPH.24 The SLF also may be damaged from mechanical stretching, metabolic derangement, or both because the SLF runs along the side of the lateral ventricle and connects broad areas of the brain that might be damaged by the diffuse metabolic derangement in iNPH.7 Thus, decreased integrity of crossing fibers also may contribute to the increased FA values in the CST. Further studies exploring the cerebral white matter damage are needed.
Visual evaluation of tractography of the CST demonstrated that most patients with iNPH (94.4%) had type B characteristics (high anisotropy and straight). In contrast, most patients with AD (72.8%) or PDD (90.9%) and most control subjects (94.7%) were of type A (low anisotropy and rough). The anisotropy, expressed as color in the tractography, followed the same trend as the FA values. Thus, the results of visual evaluation of the tractography as particular colors in patients with iNPH were consistent with the increased FA values in the tract-specific analysis. As mentioned, the CST crosses the SLF and interhemispheric fibers.22 We speculate that, in control subjects, the presence of complex microstructures in the CST, such as various crossing fibers and microscopic winding of fibers, leads to low FA values and fluctuating axial eigenvectors. These microscopic bases may underlie the macroscopic appearance of the tractography in control subjects (low anisotropy and rough). In contrast, we speculate that, in patients with iNPH, there are altered microstructures in the CST, such as stretched neural fibers with or without decreased integrity of crossing fibers and that this leads to increased FA values and less fluctuation in axial eigenvectors. These microscopic bases may underlie the macroscopic appearance of tractographic images in patients with iNPH (high anisotropy and straight).
It is often difficult to differentiate iNPH from other common diseases such as AD and PDD. CSF fluid-tap testing and external lumbar drainage testing may help to make a diagnosis of iNPH.28 However, these are invasive tests. In addition, CSF tap testing has poor sensitivity,29 and external lumbar drainage testing does not have good negative predictive values.8 Therefore, an accurate method that can identify iNPH and that preferably is noninvasive is needed. Here, we successfully differentiated patients with iNPH from patients with AD or PDD or control subjects by using tract-specific analysis and visual evaluation of tractography of the CST. These results also were confirmed in a comparison between non-iNPH subgroups and the iNPH subgroup under conditions in which all subjects had similar extents of ventricular enlargement. Thus, diffusion tensor metrics may be a more specific biomarker of iNPH than the Evans index, and DTI analysis of the CST may be a promising tool for differentiating iNPH from other diseases.
Visual evaluation of tractography is technically easier than performing tract-specific analysis. The dTV II software that we used here has been used in many DTI studies.13,30 In addition, the same algorithm as used in dTV II is integrated into the clinical MR imaging software of a major vendor (GE Healthcare). Therefore, visual evaluation of tractography is already applicable to clinical practice in certain environments and may be readily available as a screening tool for iNPH, followed by tract-specific quantitative evaluation.
This study, however, had several limitations. First, we included limited numbers of patients with AD or PDD who had Evans indexes >0.3. Further studies that include more non-iNPH patients with ventricular enlargement are necessary. Second, only limited information on the pathology of iNPH, especially in regard to the CST, is available. Thus, pathologic confirmation of altered diffusion tensor metrics is needed in a future study. Third, we did not evaluate diffusion tensor metrics in patients with iNPH who had had shunt surgery. We therefore do not know the nature of the changes in diffusion tensor isometrics in patients with iNPH after shunt surgery. Additional studies including patients who have had shunt surgery are needed.
Conclusions
This study demonstrated that FA values and axial eigenvalues were significantly greater in the CST of patients with iNPH than in control subjects. These results suggest that there are altered microstructures in the CST in patients with iNPH, presumably resulting from the mechanical pressure due to ventricular enlargement with or without a decrease in the abundance of crossing fibers. By using this method, patients with iNPH were successfully differentiated from patients with AD or PDD and control subjects. The applicability of this method should be further investigated, including more patients with secondary ventricular enlargement and nonresponders to shunt surgery.
Acknowledgments
We thank Drs Atsushi Shiraishi, Department of Emergency Medicine, and Satoru Ishibashi, Department of Neurology and Neurologic Sciences, Graduate School, Tokyo Medical and Dental University, Tokyo, Japan, for discussion. We also thank Atsuhiko Hattori, Takanori Odakura, Maki Kyono, and Ryohei Abe, Department of Radiology, Kamagaya General Hospital, for cooperation. We thank the Japanese Ministry of Education, Culture, Sports and Technology's Comprehensive Brain Science Network for providing a good environment in which to have discussions.
Footnotes
-
This study was supported (including provision of equipment or materials) by grants received in the 2009 fiscal year from Tanabe Mitsubishi Co., Novartis Co., BoelingerIngerheim Co., Takeda Chemical Industries, Eisai Co., Dainipponsumitomo Co., AstraZeneca Co., Pfizer Co., Asteras Co., GlaxoSmitKline Co., Kissei Co., Sanofi Aventis Co., Teijin Phama Co., FP Co., Banyu Pharmaceutical Co., Otsuka Pharmaceutical Co., Japan Mediphysics Co. and Tsumura Co.
Disclosures: Hidehiro Mizusawa; Speaker Bureau: Honoraria for sponsored lectures in the 2009 fiscal year: Details: Tanabemitsubishi Co.; Sanofi Aventis Co.
Indicates open access to non-subscribers at www.ajnr.org
References
- Received January 11, 2011.
- Accepted after revision January 24, 2011.
- © 2011 by American Journal of Neuroradiology